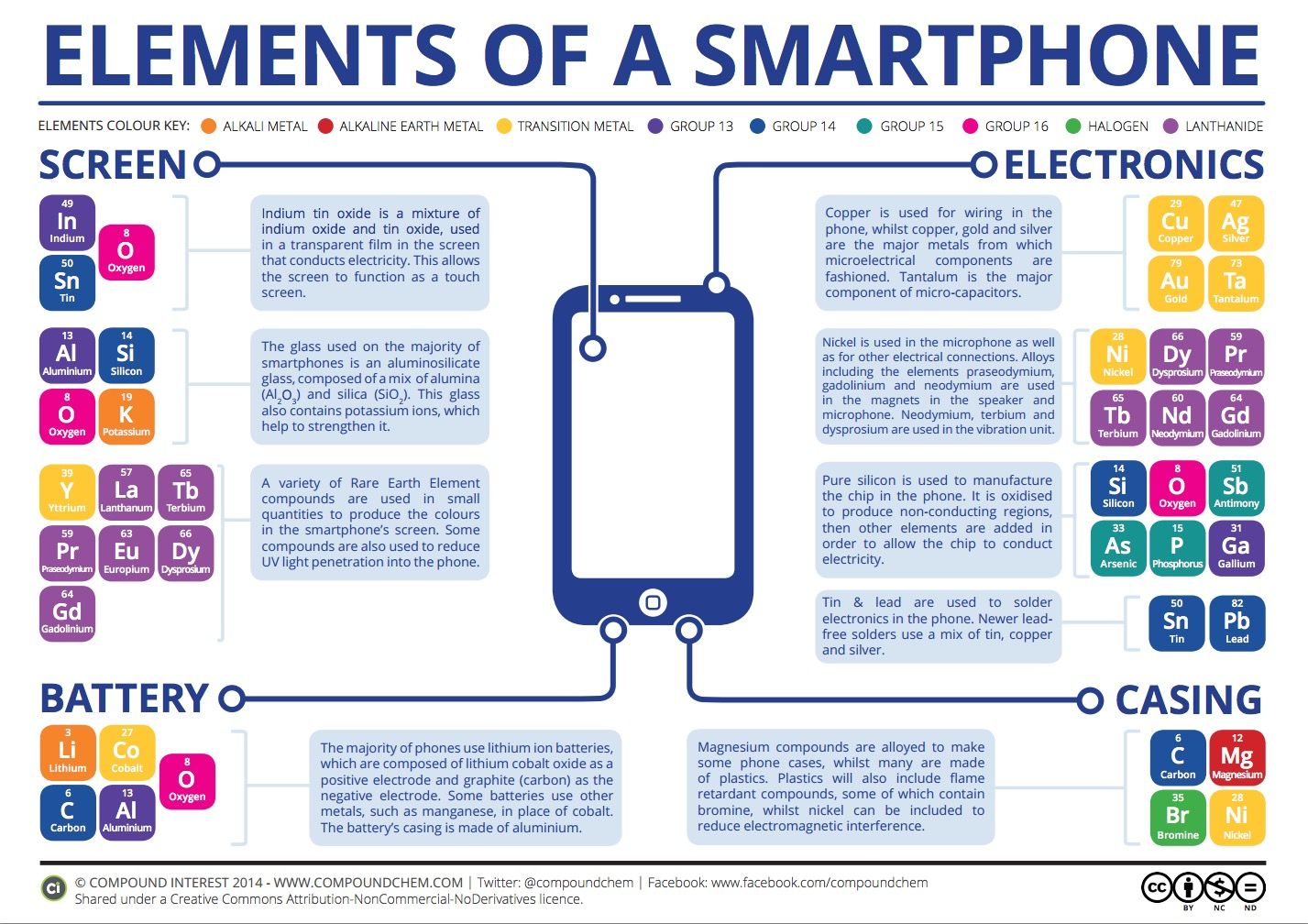
The average smartphone is a complex piece of machinery, often hiding between deceptively simple looks. But just look inside of that case and you’d see a whole mechanism of various parts all coming together to make it work.
Some interesting revelations about the secret metals and materials were recently summarized in a concise and informative video that we share with you below. In it, you’d find out that your typical smartphone contains around 300mg of silver and 30mg gold. That may not sound like much, but if you look at the whole phone industry, it combines for a whopping $2.5 billion of expenses.
You, of course, know that your phone requires a battery (and the larger, the better), but you might not have known that the battery contains elements like Lithium-cobalt oxide and Carbon/graphite. The body of the phone? A range of materials is used in it: aluminum for a lot of premium devices and plastic for less expensive ones.
Finally, you’d hear about fancy features like the new ion-strengthened glass on the iPhone 6, made by bathing the display cover and compressing the glass with potassium. Then, a compound of indium, tin, and oxygen is actually what makes it possible to touch your phone, forming a transparent film that conducts electricity. And, naturally, silicon is used for the processor. Ultimately, though, it’s how they all come together, often in miniature amounts, to make your phone tick.
Read the latest from Victor Hristov



Things that are NOT allowed:
To help keep our community safe and free from spam, we apply temporary limits to newly created accounts: